|
by Ariff BONGSO
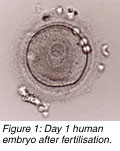
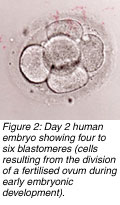
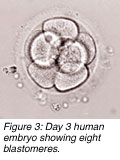
 n most in vitro fertilisation (IVF) centres, embryos are placed into a patients uterus on the second or third day at the four- or eight-cell stages, respectively. Although high pregnancy rates can result with such day-2 or day-3 transfers, some studies show that the transfer of five-day-old embryos (blastocysts) can enhance pregnancy outcome. n most in vitro fertilisation (IVF) centres, embryos are placed into a patients uterus on the second or third day at the four- or eight-cell stages, respectively. Although high pregnancy rates can result with such day-2 or day-3 transfers, some studies show that the transfer of five-day-old embryos (blastocysts) can enhance pregnancy outcome.
 There are three justifications for this statement. First, more physiological viability exists in returning a blastocyst to the uterine environment than returning an earlier cleavage-stage embryo because the uterine environment is designed to be most receptive to the four- or five-day-old human embryo. Second, only the hardiest embryos will survive in culture for five days, and third, embryo selection is better and more precise with day-5 embryos. There are three justifications for this statement. First, more physiological viability exists in returning a blastocyst to the uterine environment than returning an earlier cleavage-stage embryo because the uterine environment is designed to be most receptive to the four- or five-day-old human embryo. Second, only the hardiest embryos will survive in culture for five days, and third, embryo selection is better and more precise with day-5 embryos.
Some researchers have claimed that because higher implantation rates result from blastocyst transfers, the transfer of one blastocyst will avoid a multiple-sibling pregnancy (one side effect of IVF). Other scientists believe that appearance alone under the microscope of day-3 embryos poorly predicts future blastocyst quality and that no correlation exists between blastulation rates of cultured spare embryos and transfers on day 2 or 3 in the same patients. Additional studies report contradictory views that blastocyst transfers alone were no better than day-3 transfers in IVF patients.
 It is important to understand the physiology and behaviour of the human embryo in the first five days after fertilisation. A human embryos genetic makeup activates between day 2 and 3 of growth, following which the embryo takes full control of its own developmental fate. Until this time the mothers genome fully controls early embryonic development. If the embryo lacks some very special nutrients and other environmental conditions conducive for healthy development between day 2 and 3, "human in vitro embryonic block" occurs - the embryo ceases to grow. Thus, transferring embryos after day 2 in IVF patients should theoretically be beneficial. However, transfer done before it is clear that the embryos have not undergone the block could result in some undesirable in vitro carry-over effects from the first and second days spent in the laboratory. It is important to understand the physiology and behaviour of the human embryo in the first five days after fertilisation. A human embryos genetic makeup activates between day 2 and 3 of growth, following which the embryo takes full control of its own developmental fate. Until this time the mothers genome fully controls early embryonic development. If the embryo lacks some very special nutrients and other environmental conditions conducive for healthy development between day 2 and 3, "human in vitro embryonic block" occurs - the embryo ceases to grow. Thus, transferring embryos after day 2 in IVF patients should theoretically be beneficial. However, transfer done before it is clear that the embryos have not undergone the block could result in some undesirable in vitro carry-over effects from the first and second days spent in the laboratory.
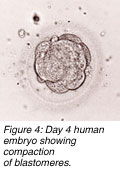
Extending culture to five days also gives embryologists time to weed out genetically inferior embryos. With continuing development of good embryos, a better selection of blastocysts for transfer remains. One study showed that IVF patients with a poor prognosis had more than 50% genetically abnormal embryos. In another study, 40% of blastocysts in extended culture showed chromosomal mosaicism (a group of disorders comprising numerical or structural chromosomal irregularities) of the inner cell mass from which the future foetus arises.
 Although extended culture has several benefits, it also involves certain challenges. Growth of human embryos to blastocysts requires stringent quality control and close monitoring of the embryos laboratory environment for five days. Embryos require a complex nourishing culture medium on days 1 and 2 and are switched to a second super-complex nourishing medium from days 3 to 5, making increased incubator space necessary for five days to avoid fluctuations of pH and osmolarity arising from frequent opening and closing of incubator doors. Although extended culture has several benefits, it also involves certain challenges. Growth of human embryos to blastocysts requires stringent quality control and close monitoring of the embryos laboratory environment for five days. Embryos require a complex nourishing culture medium on days 1 and 2 and are switched to a second super-complex nourishing medium from days 3 to 5, making increased incubator space necessary for five days to avoid fluctuations of pH and osmolarity arising from frequent opening and closing of incubator doors.
Doctors cannot guarantee all patients signed up for IVF a blastocyst transfer because blastulation rates rarely exceed 60- 70%. Older patients, who usually generate fewer eggs, have similarly fewer blastocysts. Some IVF centres have laid down specific criteria for transfer, such as availability of a minimum of four to five good embryos on day 3 so as to increase the chances of pregnancy but also to ensure availability of one or two blastocysts for transfer on day 5.
 Some centres have even proposed predictive criteria to identify as early as day 1 the best embryos to yield blastulation, implantation, and pregnancy whereas others have developed a graduated embryo-scoring system based on appearance under special microscopes, after scoring embryos for quality on days 1 to 3. Some centres have even proposed predictive criteria to identify as early as day 1 the best embryos to yield blastulation, implantation, and pregnancy whereas others have developed a graduated embryo-scoring system based on appearance under special microscopes, after scoring embryos for quality on days 1 to 3.
The IVF programme at the National University Hospital in Singapore comprises caring and dedicated embryologists with long years of experience with extended culture. They have refined the extended culture protocol to attain high pregnancy rates of at least 50-55%. Embryo transfer is carried out after the in vitro embryonic block on either day 3, 4, or 5 and not day 5 alone. The protocol virtually guarantees a patient embryo transfer on any of these days instead of targeting her for a day-5 blastocyst transfer only.
 The pregnancy rates rise linearly with day 3, 4, and 5 transfers when compared to day 2. The laboratory protocol incorporates: The pregnancy rates rise linearly with day 3, 4, and 5 transfers when compared to day 2. The laboratory protocol incorporates:
- an integrated daily egg and embryo scoring system from day 0 to 5.
- a brief two-hour exposure of sperm and egg for fertilisation (instead of overnight exposure) or the microinjection of a single sperm into an egg.
- extended culture of embryos in a small controlled environment using small plastic incubators and two-stage gas environments.
- growth of individual embryos in microdroplets of stagespecific sequential culture media.
- decision about day of transfer made by an embryologist on duty who specialises on embryo development, relying on high cumulative embryo scores.
Patients were more comfortable if told upfront that they would be targeted for day 5 transfers and that if a hint of no blastocyst production after daily embryo scoring by day 3 remained, they would still get a day 3 or 4 transfer with high pregnancy rates. In this way, no patient went home disappointed if she did not have a blastocyst transfer.
Selecting embryos that have a high implantation potential is one of the major contributory factors to improved results in an IVF programme. Embryo quality has traditionally been evaluated on the basis of speed of growth and appearance under the microscope on the day of transfer. This single evaluation lacks sensitivity and specificity in predicting pregnancy outcome, prompting IVF specialists to transfer more embryos (up to four), leading in turn to increased multiple-foetus rates.
Although it is tempting to correlate egg and embryo scoring with implantation, other variables exist beyond this screening. Thus, one parameter used in isolation to determine outcome must be viewed with caution. Other parameters such as a standardised microdroplet culture, using stage-specific culture media, a brief two-hour insemination, and a standardised ultrasound-guided embryo transfer, plus the egg and embryo scoring, all contribute to high pregnancy rates in one way or another.
For more information contact Ariff Bongso at obgbongs@nus.edu.sg
 Click here to download the full issue for USD 6.50 Click here to download the full issue for USD 6.50
|


 n most in vitro fertilisation (IVF) centres, embryos are placed into a patients uterus on the second or third day at the four- or eight-cell stages, respectively. Although high pregnancy rates can result with such day-2 or day-3 transfers, some studies show that the transfer of five-day-old embryos (blastocysts) can enhance pregnancy outcome.
n most in vitro fertilisation (IVF) centres, embryos are placed into a patients uterus on the second or third day at the four- or eight-cell stages, respectively. Although high pregnancy rates can result with such day-2 or day-3 transfers, some studies show that the transfer of five-day-old embryos (blastocysts) can enhance pregnancy outcome.
 There are three justifications for this statement. First, more physiological viability exists in returning a blastocyst to the uterine environment than returning an earlier cleavage-stage embryo because the uterine environment is designed to be most receptive to the four- or five-day-old human embryo. Second, only the hardiest embryos will survive in culture for five days, and third, embryo selection is better and more precise with day-5 embryos.
There are three justifications for this statement. First, more physiological viability exists in returning a blastocyst to the uterine environment than returning an earlier cleavage-stage embryo because the uterine environment is designed to be most receptive to the four- or five-day-old human embryo. Second, only the hardiest embryos will survive in culture for five days, and third, embryo selection is better and more precise with day-5 embryos.
 It is important to understand the physiology and behaviour of the human embryo in the first five days after fertilisation. A human embryos genetic makeup activates between day 2 and 3 of growth, following which the embryo takes full control of its own developmental fate. Until this time the mothers genome fully controls early embryonic development. If the embryo lacks some very special nutrients and other environmental conditions conducive for healthy development between day 2 and 3, "human in vitro embryonic block" occurs - the embryo ceases to grow. Thus, transferring embryos after day 2 in IVF patients should theoretically be beneficial. However, transfer done before it is clear that the embryos have not undergone the block could result in some undesirable in vitro carry-over effects from the first and second days spent in the laboratory.
It is important to understand the physiology and behaviour of the human embryo in the first five days after fertilisation. A human embryos genetic makeup activates between day 2 and 3 of growth, following which the embryo takes full control of its own developmental fate. Until this time the mothers genome fully controls early embryonic development. If the embryo lacks some very special nutrients and other environmental conditions conducive for healthy development between day 2 and 3, "human in vitro embryonic block" occurs - the embryo ceases to grow. Thus, transferring embryos after day 2 in IVF patients should theoretically be beneficial. However, transfer done before it is clear that the embryos have not undergone the block could result in some undesirable in vitro carry-over effects from the first and second days spent in the laboratory.
 Although extended culture has several benefits, it also involves certain challenges. Growth of human embryos to blastocysts requires stringent quality control and close monitoring of the embryos laboratory environment for five days. Embryos require a complex nourishing culture medium on days 1 and 2 and are switched to a second super-complex nourishing medium from days 3 to 5, making increased incubator space necessary for five days to avoid fluctuations of pH and osmolarity arising from frequent opening and closing of incubator doors.
Although extended culture has several benefits, it also involves certain challenges. Growth of human embryos to blastocysts requires stringent quality control and close monitoring of the embryos laboratory environment for five days. Embryos require a complex nourishing culture medium on days 1 and 2 and are switched to a second super-complex nourishing medium from days 3 to 5, making increased incubator space necessary for five days to avoid fluctuations of pH and osmolarity arising from frequent opening and closing of incubator doors.
 Some centres have even proposed predictive criteria to identify as early as day 1 the best embryos to yield blastulation, implantation, and pregnancy whereas others have developed a graduated embryo-scoring system based on appearance under special microscopes, after scoring embryos for quality on days 1 to 3.
Some centres have even proposed predictive criteria to identify as early as day 1 the best embryos to yield blastulation, implantation, and pregnancy whereas others have developed a graduated embryo-scoring system based on appearance under special microscopes, after scoring embryos for quality on days 1 to 3.
 The pregnancy rates rise linearly with day 3, 4, and 5 transfers when compared to day 2. The laboratory protocol incorporates:
The pregnancy rates rise linearly with day 3, 4, and 5 transfers when compared to day 2. The laboratory protocol incorporates:
 Click here to download the full issue for USD 6.50
Click here to download the full issue for USD 6.50