|
by Tan Lay Leng
 engue fever, a life-threatening infectious disease transmitted by the Aedes mosquito, occurs in crowded urban environments where the insect breeds. Healthcare providers estimate that as many as 60 million people around the world come down with dengue fever each year. Patients with dengue haemorrhagic fever (DHF) or shock syndrome, the most serious form of the disease, need immediate treatment because they may die if untreated or if treatment is delayed. engue fever, a life-threatening infectious disease transmitted by the Aedes mosquito, occurs in crowded urban environments where the insect breeds. Healthcare providers estimate that as many as 60 million people around the world come down with dengue fever each year. Patients with dengue haemorrhagic fever (DHF) or shock syndrome, the most serious form of the disease, need immediate treatment because they may die if untreated or if treatment is delayed.
According to the Singapore Ministry of Health (MOH), 1,355 cases of dengue fever occurred in Singapore in 1999. Of these cases, 30 patients contracted DHF, and among them, 5 died. The number of dengue fever cases decreased to 673 in 2000, with no death resulting. The highest incidence occurred in 1998, with 5,258 cases and 1 death.
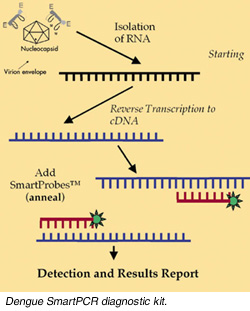 Singapore-based Asia-Pacific (AP) Genomics, a biotechnology startup company, is developing a new diagnostic kit called SmartPCR which aims to speed up the diagnosis of the potentially fatal disease (see figure). Applying pioneering technology by Associate Professor Vincent Chow of the National University of Singapore's Department of Microbiology, who designed type-specific dengue diagnostic primers, the company hopes to come up with a DNA-based diagnostic test kit that will quickly confirm infection and identify the specific type of dengue virus causing that infection, including the type II virus that leads to DHF (see box story). Singapore-based Asia-Pacific (AP) Genomics, a biotechnology startup company, is developing a new diagnostic kit called SmartPCR which aims to speed up the diagnosis of the potentially fatal disease (see figure). Applying pioneering technology by Associate Professor Vincent Chow of the National University of Singapore's Department of Microbiology, who designed type-specific dengue diagnostic primers, the company hopes to come up with a DNA-based diagnostic test kit that will quickly confirm infection and identify the specific type of dengue virus causing that infection, including the type II virus that leads to DHF (see box story).
Early diagnosis would also help public health organisations deal quickly with issues of mosquito breeding, population testing, and containment of disease spread, ultimately achieving disease prevention.
Presently, diagnostic approaches to dengue are based on clinical signs such as fever, which often lead to misdiagnosis or even death when patients do not start treatment in time, notes Dr Sharan Sambhi, who heads the project-management team at AP Genomics. The current test usually takes about a week to determine the patient's immune response, before it can confirm whether a person bitten by a mosquito has contracted dengue; the new method can detect the virus in a patient's blood in as short as three days.
Immunodiagnostic tests now available measure the antibodies triggered by infection and usually indicate a negative result until well after a fever has developed. On the other hand, DNA-based testing has the potential to confirm dengue within three to five days of onset of fever by detecting the live virus circulating in the blood. Immunologically-based tests generally come back negative during most of this period and only start showing positive results towards the tail-end of the viraemic phase, that is, beyond the seventh day.
In the DNA-based SmartPCR test, a scientist puts the patient's infected blood sample through a polymerase chain reaction (PCR) machine, which replicates the virus' genetic material. The test looks for DNA specific to the virus. The assay, which takes several hours, can also identify - if the DNA markers are present - which of the four different types of dengue virus a person has contracted.
The scientists can adapt the test to speed up the confirmation of whether field-caught mosquitoes carry the dengue virus and identify the type involved. The Ministry of the Environment in Singapore has already validated the NUS technology. The Institute for Medical Research, which is under Malaysia's Ministry of Health, will oversee the testing for field validation of the dengue diagnostic technology on patients.
AP Genomics plans to make the SmartPCR diagnostic kit available in the third quarter of this year. Soon after, the company expects to release DNA-based tests for malaria and other tropical diseases as well as sexually transmitted diseases.
For more information contact Hazelle Lam at hazelle.lam@apgenomics.com or Assoc Prof Vincent Chow at micctk@nus.edu.sg.
|
Probing for Mosquito-Borne Viruses
Associate Professor Vincent Chow at the Department of Microbiology, NUS, has designed primers or probes for use in polymerase chain reaction (PCR) DNA sequencing and nucleic acid hybridisation. A primer is a segment of DNA complementary to a given DNA sequence to initiate replication by DNA polymerase. Consensus primers are conserved primers capable of binding to nucleic-acid sequences conserved among different flaviviruses (mosquito-transmitted viruses).
In a process called reverse transcription PCR (RT-PCR), a pair of consensus primers, based on the genes of dengue viruses and other related flaviviruses, can amplify the virus; this means health professionals can adapt the technique to test for the Japanese encephalitis, West Nile, and yellow fever viruses.
Chow has also developed four type-specific primers based on the genes of the four different types of dengue virus. The combination of the consensus and type-specific primers can be used for the rapid and sensitive detection of each specific dengue virus type. The invention of these primers has been protected by patents in the United Kingdom.
Chow has successfully tested the primers on laboratory strains of dengue and other flaviviruses, as well as on clinical specimens. RT-PCR with these primers has proved to be superior to classical virus isolation or serology for the early diagnosis of dengue fever, within three to five days of the onset of the fever that occurs early in the disease process. Furthermore, Chow's team and the Vector Control and Research Department, Ministry of the Environment, Singapore have adopted the technique for regular monitoring of dengue viruses in Singapore field-caught Aedes mosquitoes. The conserved primers have been successfully used for the identification of the emerging West Nile-like flavivirus responsible for the 1999 outbreak of encephalitis in New York.
In addition to type-specific primers for dengue, Chow has also developed type-specific primers for Japanese encephalitis - a disease of major concern in some countries in East Asia where dengue is also endemic, such as China, Thailand, and Vietnam.
|
|


 engue fever, a life-threatening infectious disease transmitted by the Aedes mosquito, occurs in crowded urban environments where the insect breeds. Healthcare providers estimate that as many as 60 million people around the world come down with dengue fever each year. Patients with dengue haemorrhagic fever (DHF) or shock syndrome, the most serious form of the disease, need immediate treatment because they may die if untreated or if treatment is delayed.
engue fever, a life-threatening infectious disease transmitted by the Aedes mosquito, occurs in crowded urban environments where the insect breeds. Healthcare providers estimate that as many as 60 million people around the world come down with dengue fever each year. Patients with dengue haemorrhagic fever (DHF) or shock syndrome, the most serious form of the disease, need immediate treatment because they may die if untreated or if treatment is delayed.
 Singapore-based Asia-Pacific (AP) Genomics, a biotechnology startup company, is developing a new diagnostic kit called SmartPCR which aims to speed up the diagnosis of the potentially fatal disease (see figure). Applying pioneering technology by Associate Professor Vincent Chow of the National University of Singapore's Department of Microbiology, who designed type-specific dengue diagnostic primers, the company hopes to come up with a DNA-based diagnostic test kit that will quickly confirm infection and identify the specific type of dengue virus causing that infection, including the type II virus that leads to DHF (see box story).
Singapore-based Asia-Pacific (AP) Genomics, a biotechnology startup company, is developing a new diagnostic kit called SmartPCR which aims to speed up the diagnosis of the potentially fatal disease (see figure). Applying pioneering technology by Associate Professor Vincent Chow of the National University of Singapore's Department of Microbiology, who designed type-specific dengue diagnostic primers, the company hopes to come up with a DNA-based diagnostic test kit that will quickly confirm infection and identify the specific type of dengue virus causing that infection, including the type II virus that leads to DHF (see box story).